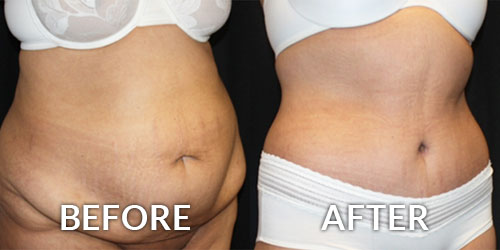
Do you have questions about a Mommy Makeover?
Motherhood is a beautiful experience. Yet, while mothers adore their children, they are often left feeling unhappy with the ways pregnancy and childbirth have changed their bodies. Loss of volume in the breasts, sagging skin, stubborn fat and weak or separated abdominal muscles are often the results of having one […]
Read More